 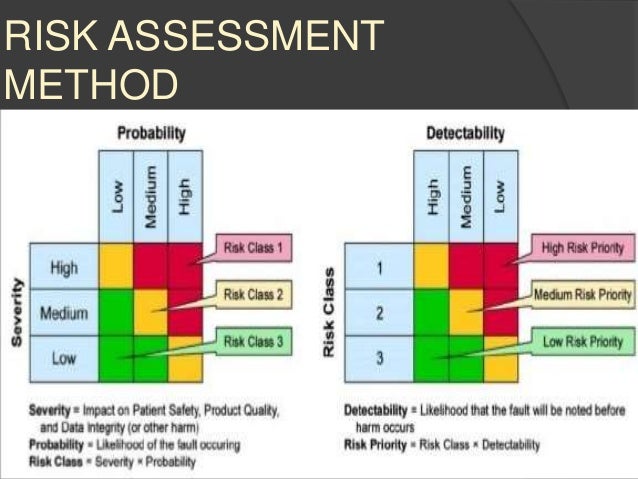
Figure 2 summarises the steps for online monitoring systems as category 5 custom application from validation planning to validation reporting. Each level on the right-hand side also verifies the same level specification steps on the left. Figure 1 shows specification steps on the right-hand side and testing, qualification and verification steps on the left-hand side. The GAMP5 V scheme summarises all specification and testing activities. The graphic is known as the GAMP5 V scheme. The specification steps indicated on the right-hand side meet the verification steps on the left-hand side. Are more detailed risk assessments required?įor the functional risk assessment, identify and define the following points:Īs you can see in Figure 1, the project phase covers several activities from planning to reporting.What is the overall impact of the system?.What are the overall risks to the business?.There are two stages here: initial risk assessment even before forming the user requirement specification (URS) and functional risk assessment at the stages of specification such as function, design, and module specifications.įor the initial risk assessment, consider the following points: To be able to apply proper GAMP5 requirements, a risk-based approach for a custom application should be maintained. These platforms feature complex structures with data modules, programmable logic controllers with custom ladder logics and custom firmware to meet client’s specific requirements. When talking about monitoring software, these are all configured systems (category 4) and even custom systems (category 5). Figure 1 also shows the typical use of risk-based decision-making throughout the life cycle. The risk-based approachĪppropriate risk management processes should be followed throughout the life cycle to manage identified risks and to determine the rigour and extent of the activities required at each phase of the life cycle. It may be suitable to delegate many of the described activities to suppliers, subject to satisfactory supplier assessment and control measures. As per GAMP5, suppliers of products and services should be involved as appropriate throughout the life cycle. The life cycle for any system consists of four major phases: concept, project, operation, and retirement. The life cycle approach is also described in GAMP5, as shown in Figure 1 (see next page). That’s why there is no straight line from A to Z for computerised system validation. The life cycle approachīecause the entire system lives as a concept, parameters, estimates and/or requirements are changing as the system lives. It’s not because they are in fashion in today's cleanroom markets both really serve for appropriate system design and implementation. When it comes to validation, two approaches are very critical: the risk-based and life cycle approach. As per categorisation, complexity and risk increased, as shown in the sequence below:Ĭustom (Category 5) > Configured (Category 4) > Non-configured (Category 3) > Infrastructure (Category 1) The categories are useful when applied in conjunction with other risk management tools, and with consideration of complexity and size of the system. 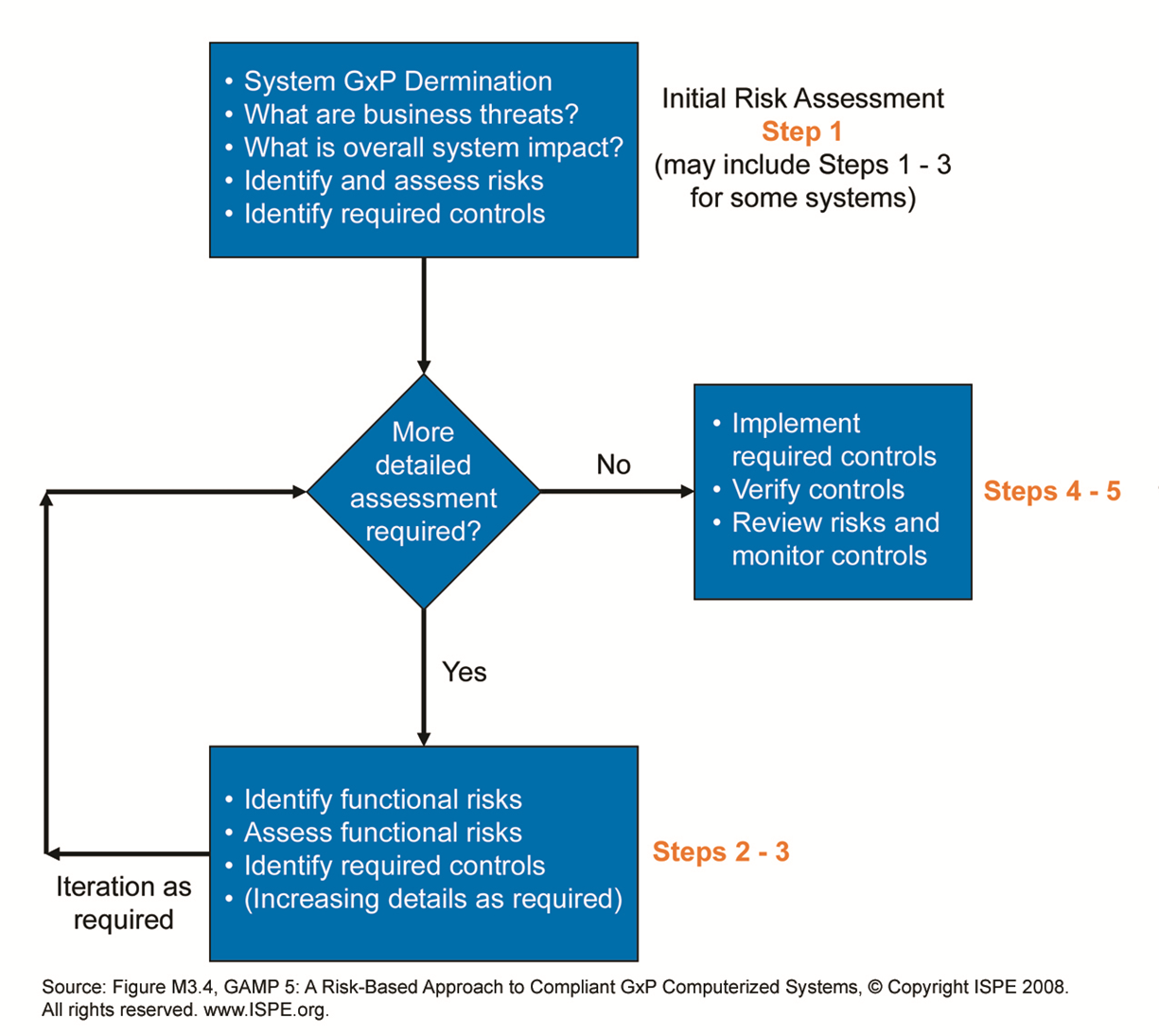
When coupled with risk assessment and supplier assessment, categorisation can be part of an effective quality risk management approach. The table below gives more details about the categories and their intended use. Moreover, the system categorisation helps guide the efficient writing of documentation, including specifications, test scripts and everything in between. The document's categorisation assists in selecting appropriate life cycle activities and documentation from category 1 “infrastructure software” up to category 5 “custom applications”. Volume 5 of the GAMP (GAMP5) was published in 2008 by the ISPE task team alongside subject matter experts from field and steering committees from various countries. GAMP is an acronym for Good Automated Manufacturing Practice, and it is both a technical subcommittee of the International Society for Pharmaceutical Engineering (ISPE) and a set of guidelines for manufacturers and users of automated systems in the pharmaceutical industry for a risk-based approach to computer system validation. Following on from the guide to designing a cleanroom monitoring system, published in the September issue of Cleanroom Technology, this article is set up to cover the critical aspects involved in the installation of a monitoring system under the validation steps as per the guidelines of the ISPE GAMP5. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
